Multiple Choice
Identify the
choice that best completes the statement or answers the question.
|
|
|
1.
|
Rings are known to exist around which of the following planets:
A) | Saturn and Neptune | B) | Saturn | C) | Saturn and
Jupiter | D) | all of the Jovian planets |
|
|
|
2.
|
Which of the following diagrams correctly displays the bonding between S and Cl
in SCl2?
A) | S—Cl | B) | S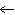 Cl Cl | C) | S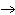 Cl Cl | D) | all of the
above |
|
|
|
3.
|
The existence of Neptune was proposed
A) | because of its effect on Pluto’s orbit | B) | because of its
effect on Uranus’s orbit | C) | on the basis of photographs | D) | because of
ground-based direct observations |
|
|
|
4.
|
The Great Red Spot on Jupiter is probably
A) | a hole in the clouds | B) | a storm | C) | an optical
illusion | D) | a continent |
|
|
|
5.
|
The correct name for N2O is
A) | dinitrogen oxide | B) | nitrogen oxide | C) | dinitrogen
monoxide | D) | nitrogen monoxide |
|
|
|
6.
|
Which of the following is NOT a physical change?
A) | condensing of steam | B) | burning of gasoline | C) | freezing
water | D) | crushing salt |
|
|
|
7.
|
Which of the following is a chemical property?
A) | melts at 73 °C | B) | has a high
density | C) | good reflector of light | D) | decomposes upon
heating |
|
|
|
8.
|
The name for compound K3N is
A) | potassium nitride | B) | tripotassium nitrogen | C) | tripotassium
nitride | D) | potassium nitrate |
|
|
|
9.
|
The planet that is most similar to Earth with respect to mass, diameter and
density is
A) | Venus | B) | Mars | C) | Jupiter | D) | Mercury |
|
|
|
10.
|
A substance that increase the rate of a reaction but is not consumed in the
reaction is called
A) | a redox agent | B) | a reactant | C) | a
catalyst | D) | a product |
|
|
|
11.
|
Which of the following is the correct name for Na2S?
A) | disodium sulfate | B) | sodium sulfide | C) | disodium
sulfide | D) | sodium sulfate |
|
|
|
12.
|
Most of the mass of the solar system is in the
A) | Jovian planets | B) | comets | C) | terrestrial
planets | D) | sun |
|
|
|
13.
|
The total number of atoms present in the formula
Al2(SO4)3 is
|
|
|
14.
|
Which of the following elements normally exists as a diatomic molecule?
A) | sulfur | B) | chlorine | C) | argon | D) | carbon |
|
Short Answer
|
|
|
15.
|
Balance the equation shown below:
|